Research - International Journal of Medical Research & Health Sciences ( 2021) Volume 10, Issue 9
Two Cases of Autoimmune Haemolytic Anaemia in Patients Infected with HIV
Amol Jogdand, Arun Tyagi*, Marcia Waran, AK Srivastava and AB KhareArun Tyagi, Department of Medicine, DVVPF’s Medical College, Vadgaon Gupta, Post-MIDC, Ahmednagar, Maharashtra, India, Email: aruntyagidr@gmail.com
Received: 18-Aug-2021 Accepted Date: Sep 23, 2021 ; Published: 30-Sep-2021
Abstract
Patients with HIV/AIDS are prone to develop multiple complications and comorbidities. The hematological complications including cytopenias, lymphoma, multiple myeloma, bone-marrow dysplasia, and coagulation disorders are common. Anemia is the most common cytopenia and develops in almost all patients with HIV/AIDS. Autoimmune Haemolytic Anaemia (AIHA) is a rare cause of anemia in persons infected with HIV. AIHA usually occurs at an advanced stage of HIV disease. The mortality in AIHA is about 11%. RBC autoantibodies are frequently detected after blood transfusion in patients with HIV/AIDS, necessitating screening of patients with anemia for autoantibodies. AIHA may lead to life-threatening thromboembolic complications. Therefore, RBC transfusions should be limited only to cases with severe anemia. Steroids are the first line of treatment. Rituximab, immunosuppressants, and finally, splenectomy are the therapeutic options for steroid-refractory patients. We report two female patients infected with HIV/AIDS who presented with severe anemia and positive Coomb’s Test. Both patients were successfully managed as AIHA with blood transfusion, steroids, and immunosuppressants.
Keywords
Anaemia, Haemolytic, Autoimmune, HIV/AIDS, Rituximab, Immunosuppressive agents
Introduction
Human Immunodeficiency Virus (HIV/AIDS) is a major health challenge in developing countries like India. There were an estimated 2.348 M (1.798-3.098 M) people living with HIV/AIDS (PLHIV) in India in 2019. Out of these, the highest number 0.396 M, hails from the state of Maharashtra [1]. PLHIV are prone to develop multiple complications and comorbidities. Some complications are the direct result of long-term infection, whereas others are the indirect result of aging, Antiretroviral Therapy (ART), or other patient factors [2]. The hematological complications are common in HIV/AIDS. These complications may occur at any stage of infection due to direct cytopathic effects of the virus, secondary to various infections and neoplasms, drug toxicity, and/or immune-mediated destruction of cells [3]. Hematological complications include cytopenias, lymphoma, multiple myeloma, bone marrow dysplasia, and coagulation disorders. Cytopenias may result either from bone marrow production defects or increased peripheral loss or destruction of blood cells. Anemia is the most common cytopenia and develops in almost all (up to 95%) of the PLHIV in their lifetime [4]. The yearly incidence of developing anemia increases with disease progression, affecting 3% of all cases with asymptomatic HIV infection, 12% of those with CD4 cell counts <200/μl, and 37% of patients having an AIDS-related illness [5,6]. Anemia in HIV/AIDS is associated with decreased survival, increased disease progression, and deterioration in the quality of life [5].
Autoimmune Haemolytic Anaemia (AIHA) is a rare cause of anemia in persons infected with HIV [7,8]. The incidence of AIHA in PLHIV was found to be 3.06% [9]. Anemia can be severe and may require repeated blood transfusions. These patients are at an increased risk of potentially fatal thromboembolism after blood transfusions [10]. Therefore, it is pertinent to screen PLHIV with anemia for AIHA. We report two cases of AIHA recently managed in our tertiary care teaching hospital located in the suburban area of Western Maharashtra.
Case Report
Case 1
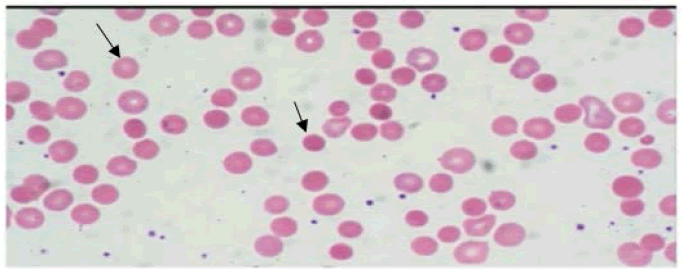
50-year-old female, a known case of HIV/AIDS for 10-years on regular ART consisting of Tenofovir, Lamivudine, and Efavirenz (TLE), was admitted with a history of exertional, gradually progressive, dyspnoea, easy fatiguability, and anorexia for two months. There was no history of fever, cough, or bleeding from any site. She denied a history of blood transfusions or any other major illness in past. Examination revealed a thin-built, pale woman who had mild icterus; the rest of the general examination and vital parameters were normal except for mild tachypnoea. Cardiovascular examination revealed a grade III/VI haemic murmur. The spleen was palpable 3 cm below the left costal margin, soft, and non-tender. Fundus examination showed flame hemorrhages. Investigation revealed severe anemia (Haemoglobin 4.2 gm/dl), normal leukocyte, and platelet count. The ESR was 79 mm at the end of 1st hour. Serum Lactate Dehydrogenase (LDH) level was 1299 U/l. The reticulocyte and corrected reticulocyte count were 2% and were 0.24% respectively. Total bilirubin was 2.97 mg/dl (Indirect 1.47 mg/dl). CD4 count was 659/mm3. Other biochemical parameters were normal. The Antinuclear Antibody (ANA) was negative. The Direct Coomb’s Test (DAT) was positive. Abdominal ultrasonography showed splenomegaly. Peripheral blood smear showed microcytic hypochromic anemia with anisocytosis and occasional spherocytes (Figure 1). Bone marrow aspiration was suggestive of dimorphic anemia.
She was diagnosed as a case of AIHA and managed with three units of packed red cells transfusion and started on oral Prednisolone 40 mg once a day and Azathioprine (AZA) 100 mg once a day along with ART and haematinics. The ART was continued, and she was reviewed in medical outpatient every month. Her blood counts remained within the normal range on prednisolone and AZA but she deteriorated once steroids were tapered off after three months and her hemoglobin dropped to 3.6 g/dl. She was transfused with 4 units of packed red cells. managed with four doses of Rituximab 500 mg infused in 500 ml normal saline over 6 h given weekly. She responded well and tolerated RTX without any major side effects. Her CD4 count in December 2020 was 1926/mm3. The last dose of RTX was given in January 2021. The steroids were finally tapered off. She is now asymptomatic and her latest hemoglobin was 12.3 g/dl.
Case 2
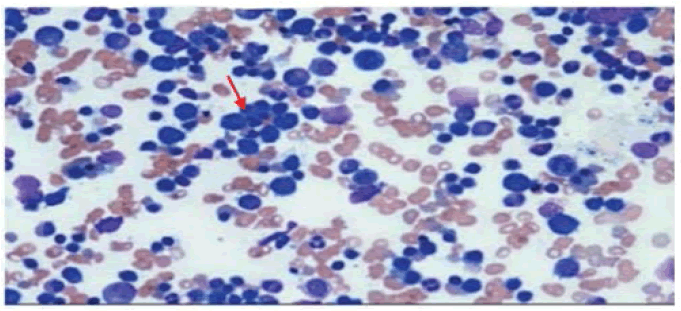
44-year-old female, case of HIV/AIDS and Diabetes Mellitus (T2DM) for 10 years on regular ART consisting of TLE, and Oral Hypoglycemic Drugs (OHA). She reported progressive exertional dyspnea, easy fatigability, and anorexia of one-month duration. There was no history of fever, cough with expectoration, bleeding from any site, or blood transfusion. Clinically, she was average built, had marked pallor and mild icterus. The rest of the general examination was unremarkable. Her pulse was 100/min, regular, high volume. She had a postural drop of 20 mm of Hg in systolic blood pressure. CVS examination revealed a grade III/VI haemic murmur. The spleen was just palpable, soft, and nontender. Investigations revealed severe anemia (Hb 3.5 gm/dl). Serum LDH level was high (1200 U/l). Total bilirubin was 2.2 mg/dl (indirect bilirubin 1.8 mg/dl). The Reticulocyte count was 2.0% and the corrected reticulocyte count was 0.18%. Blood glucose fasting was 140 mg/dl, post-prandial 220 mg/dl, and HbA1c level was 7.6%. Peripheral blood smears showed anisopoikilocytosis, microcytes, and occasional spherocytes. Bone marrow aspiration was suggestive of erythroid hyperplasia and lymphoid follicles in 6 to 7 regions (Figure 2). The DAT was positive. The absolute CD4 count was 460 cells/ml. ANA was negative. Ultrasound abdomen was suggestive of splenomegaly (16 cm).
The patient was diagnosed to have AIHA and was managed with packed RBCs transfusion and Cyclosporine 100 mg twice a day along with haematinics and glycaemic control. After one month of treatment with cyclosporine, Hb increased to 9.6 g/dl. Cyclosporine was continued in a dose of 100 mg twice a day for 06 months, 50 mg twice a day for a further six months, and 50 mg daily thereafter. Her hemoglobin has remained in the normal range on cyclosporine (latest 12.9 g/dl) and she is asymptomatic.
Discussion
Autoimmune Haemolytic Anaemia (AIHA) is an acquired, heterogeneous group of diseases mediated by autoantibodies and/or complement, together with activated macrophages, T-lymphocytes, and cytokines directed against RBCs causing premature erythrocyte destruction [11]. AIHA is defined as the increased destruction of RBC in the presence of anti-RBC autoantibodies and/or complement. Hemolysis in AIHA is mainly extravascular in the spleen and other reticuloendothelial tissues [12]. The autoantibodies may bind to the RBCs at normal temperature (37°C), the warm antibodies that are seen in 60%-90% cases of AIHA, and cold antibodies that react at around 30°C. AIHA is therefore categorized as per the type of autoantibodies, warm (wAIHA), cold (cAIHA), mixed, and drug-induced AIHA [13]. Warm AIHA is seen mostly in viral infections like HIV/AIDS, VZV, CMV, HCV, SARS-CoV-2 [14]. The incidence of AIHA is 1.77 cases per 100,000 per year. wAIHA is the most common form and accounts for about 2/3 of cases [15]. HIV/AIDS infection may induce the production of autoantibodies, owing to molecular mimicry, and causes immune dysregulation facilitating in development of autoimmune diseases. AIHA usually occurs at an advanced stage of HIV disease and carries a mortality of about 11% [16,17].
A positive Direct Antiglobulin Test (DAT) confirms the presence of immunoglobulins (most often of the IgG class, sometimes IgM and IgA and/or complement-usually C3d) attached to erythrocytes and is used for diagnosis of AIHA. However, diagnosis of AIHA may be tricky in cases of HIV/AIDS because DAT can give false-positive results in HIVinfected patients [18]. As seen in our patients, reticulocytopenia, in presence of bone marrow erythroid hyperplasia, may occur due to in vivo hemolysis of reticulocytes and adds to the confusion in the diagnosis of AIHA [19]. Patients with HIV and AIHA are at an increased chance of potentially fatal thromboembolism when they receive RBC transfusion; it is therefore required to screen HIV patients with anemia for AIHA [20].
AIHA is more common in women compared to men. Our patients were also females harboring the HIV infection for more than 10 years. O’Connell, et al. found a 71% higher prevalence of anemia among women compared to men [21]. The clinical course of AIHA may be episodic or chronic and can vary from mild to life-threatening. RBC autoantibodies are quite frequently detected after blood transfusion. Yet, clinically significant AIHA is rare but may be life-threatening, and therefore, RBC transfusions should be limited only to critical cases with severe anemia (Haemoglobin <6 g/dl) and/or hemodynamically unstable patients [22,23]. The mainstay of the treatment is centered on the inhibition of autoantibody production by mono-or combination therapy.
The first line of treatment for AIHA in HIV is corticosteroids or immunoglobulins. Prednisolone is usually started at the dose of 1.0 mg/kg/day-1.5 mg/kg/day. Optionally, an equivalent dose of methylprednisolone can be administered intravenously [8]. About 80% of patients show improvement within 2-3 weeks. After stabilization of hemoglobin, prednisone should be gradually tapered off. AIHA patients should be treated for a minimum of three or four months with low-dose prednisone (≤ 10 mg/day) [24]. The response to steroids is usually favorable but it is important to note that such patients may have to worsen their HIV status due to side effects of steroids [23]. We managed our first case with steroids alone in beginning and with azathioprine when the anemia worsened on reduction of the dose of prednisolone. We had to opt for AZA over RTX as the second choice because of financial constraints. However, the patient relapsed even on AZA and finally was managed with RTX.
The second-line therapy for AIHA, a combination of RTX, a chimeric human IgG1-κ monoclonal antibody against the protein CD20, and steroids give a better response than corticosteroid monotherapy. RTX is administered usually at a dose of 375 mg/m2 once weekly for 4-weeks. The RTX treatment is more effective and gives a response rate of 80%-90% [25,26]. Immunosuppressive drugs like azathioprine, cyclophosphamide, and cyclosporine were used as second-line therapy before RTX was introduced in the therapy of AIHA. We managed our second case with cyclosporine instead of steroids because of co-existing diabetes mellitus and fear of upsetting her glycaemic control. Immunosuppressant drugs induce remission in about 75% of WAIHA patients. The patients on immunosuppressive therapy need to be monitored closely for bone-marrow depression, renal and hepatic dysfunction, and most importantly for severe opportunistic infections [27]. Splenectomy is the third-line option for the treatment of WAIHA in young patients and females who wish to become pregnant. Splenectomy shows the response in about 70%-80% and cures about 20% of cases. Splenectomy is associated with complications related to surgery, infections, and thrombotic episodes. Infections after splenectomy carry a mortality of around 50% even after pre-operative vaccination against pneumococci, meningococcal, and Hemophilus [27]. The splenectomy rate is therefore progressively decreasing in recent years. Table 1 shows treatment options available for the treatment of AIHA [28]. Supportive treatment including vitamin supplementation, recombinant erythropoietin, thrombosis prophylaxis, and the prevention and treatment of infections is essential in patients of AIHA. Blood transfusion is risky and is indicated only in patients with severe anemia (Hb below 6 g/dl). The smallest volume of blood required to maintain adequate oxygen delivery should be transfused at the rate of less than 1 mL/kg/hour. Usually, the universal RBC donor (‘O’ Rh-) group is transfused because cross-matching in presence of warm autoantibodies complicates routine pre-transfusion testing and demands the use of specialized techniques [29,30]. The newer therapies are targeted at B and T lymphocytes, complement cascade, spleen tyrosine kinase, and the neonatal crystallizable fragment of the receptor (FcRn) present on endothelial cells and macrophages [31].
| Drug | Dose | Response to Treatment | ||
|---|---|---|---|---|
| Response Time | Initial, % | Long-term, % | ||
| Prednisone | 1.0 mg/kg-2.0 mg/kg | 1-3 weeks | 70-80 | ≤ 20 |
| Dexamethasone | (4 × 40) mg/d, 1-4 cycles/2-4 weeks | Few days | 70-80 | ≤ 20 |
| Azathioprine | 2 mg/kg-4 mg/kg | 1-3 months | 60-70 | 60-70 |
| Mycophenolate mofetil | 1 mg/kg-2 mg/kg | 2-4 weeks | 60-70 | 60-70 |
| Cyclophosphamide | 1 mg/kg-2 mg/kg | 2-4 weeks | 80-90 | 80-90 |
| Rituximab | (4 × 375) mg/m2/week | 1-8 weeks | 60-70 | ≤ 20 |
| Splenectomy | -- | Few days | <50 | <50 |
Conclusion
AIHA is a rare cause of anemia in HIV/AIDS thought to be secondary to immune dysregulation. The HIV/AIDS patients with anemia should be screened for autoantibodies because AIHA leads to life-threatening thromboembolic complications. Steroids are the first line of treatment. Steroid refractory cases, respond well to RTX and immunosuppressive therapy. Splenectomy should be the last option for the management of AIHA.
Declarations
Conflicts of Interest
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
References
- HIV Facts & Figures | National AIDS Control Organization. http://naco.gov.in/hiv-facts-figures
- Chu, Carolyn, and Peter A. Selwyn. "Complications of HIV infection: A systems-based approach." American Family Physician, Vol. 83, No. 4, 2011, pp. 395-406.
- Coffey, Susa, and Oliver Bacon. "Comprehensive, up-to-date information on HIV/AIDS treatment and prevention from the University of California San Francisco." 2019.
- Bhardwaj, Subhash, et al. "Hematologic derangements in HIV/AIDS patients and their relationship with the CD4 counts: A cross-sectional study." International Journal of Clinical and Experimental Pathology, Vol. 13, No. 4, 2020, pp. 756-63.
- Redig, Amanda J., and Nancy Berliner. "Pathogenesis and clinical implications of HIV-related anemia in 2013." Hematology 2013, the American Society of Hematology Education Program Book, Vol. 2013, No. 1, 2013, pp. 377-81.
- Meidani, Mohsen, et al. "Prevalence, severity, and related factors of anemia in HIV/AIDS patients." Journal of Research in Medical Sciences: The Official Journal of Isfahan University of Medical Sciences, Vol. 17, No. 2, 2012, pp. 138-42.
- Liebman, Howard A., and Ilene C. Weitz. "Autoimmune hemolytic anemia." The Medical Clinics of North America, Vol. 101, No. 2, 2016, pp. 351-59.
- Zanella, Alberto, and Wilma Barcellini. "Treatment of autoimmune hemolytic anemias." Haematologica, Vol. 99, No. 10, 2014, pp. 1547-54.
- Olayemi, E., O. A. Awodu, and G. N. Bazuaye. "Autoimmune haemolytic anemia in HIV patients." Annals of African Medicine, Vol. 7, No. 2, 2008, pp. 72-76.
- Zoller, Bengt, et al. "Autoimmune diseases and venous thromboembolism: A review of the literature." American Journal of Cardiovascular Disease, Vol. 2, No. 3, 2012, pp. 171-83.
- Jager, Ulrich, et al. "Diagnosis and treatment of autoimmune hemolytic anemia in adults: Recommendations from the First International Consensus Meeting." Blood Reviews, Vol. 41, 2020, p. 100648.
- Saif, Muhammad Wasif. "HIV-associated autoimmune hemolytic anemia: An update." AIDS Patient Care and STDs, Vol. 15, No. 4, 2001, pp. 217-24.
- Seve, Pascal, et al. "Autoimmune hemolytic anemia: Classification and therapeutic approaches." Expert Review of Hematology, Vol. 1, No. 2, 2008, pp. 189-204.
- Hill, Quentin A., et al. "The diagnosis and management of primary autoimmune haemolytic anaemia." British Journal of Haematology, Vol. 176, No. 3, 2017, pp. 395-411.
- Hansen, Dennis Lund, et al. "Increasing incidence and prevalence of acquired hemolytic anemias in Denmark, 1980-2016." Clinical Epidemiology, Vol. 12, 2020, pp. 497-508.
- Adewumi, A. Adediran, et al. "Prevalence of HIV-related autoimmune haemolytic anaemia in Lagos, Nigeria." Nigerian Medical Journal: Journal of the Nigeria Medical Association, Vol. 55, No. 1, 2014, pp. 63-66.
- Telen, Marilyn J., Kenneth B. Roberts, and John A. Bartlett. "HIV-associated autoimmune hemolytic anemia: Report of a case and review of the literature." JAIDS Journal of Acquired Immune Deficiency Syndromes, Vol. 3, No. 10, 1990, pp. 933-37.
- Barros, Melca MO, Morris A. Blajchman, and Jose O. Bordin. "Warm autoimmune hemolytic anemia: Recent progress in understanding the immunobiology and the treatment." Transfusion Medicine Reviews, Vol. 24, No. 3, 2010, pp. 195-210.
- Conley, C. Lockard, et al. "Autoimmune hemolytic anemia with reticulocytopenia and erythroid marrow." New England Journal of Medicine, Vol. 306, No. 5, 1982, pp. 281-86.
- Balogun, Taiwo Modupe, et al. "The pattern of haematological changes in the baseline blood cell counts and the CD4⁺ T lymphocyte levels among antiretroviral therapy naïve adult HIV positive patients in a Nigerian hospital." Open Journal of Blood Diseases, Vol. 10, No. 3, 2020, pp. 77-88.
- O'Connell, Casey, and Alexandra M. Levine. "Managing anemia in HIV-positive women." Women’s Health, Vol. 2, No. 1, 2006, pp. 159-65.
- Sigler, Erica, et al. "Clinical significance of serologic markers related to red blood cell autoantibodies production after red blood cell transfusion-severe autoimmune hemolytic anemia occurring after transfusion and alloimmunization: Successful treatment with rituximab." Transfusion, Vol. 49, No. 7, 2009, pp. 1370-74.
- Park, Sang Hyuk, Won-Ho Choe, and Seog-Woon Kwon. "Red blood cell transfusion in patients with autoantibodies: Is it effective and safe without increasing hemolysis risk?" Annals of Laboratory Medicine, Vol. 35, No. 4, 2015, pp. 436-44.
- Jaime-Perez, Jose Carlos, et al. "Treatment of autoimmune hemolytic anemia: Real world data from a reference center in Mexico." Blood Research, Vol. 54, No. 2, 2019, pp. 131-36.
- Go, Ronald S., Jeffrey L. Winters, and Neil E. Kay. "How I treat autoimmune hemolytic anemia." Blood, The Journal of the American Society of Hematology, Vol. 129, No. 22, 2017, pp. 2971-79.
- Chao, Shih-Hsuan, et al. "Efficacy and safety of rituximab in autoimmune and microangiopathic hemolytic anemia: A systematic review and meta-analysis." Experimental Hematology & Oncology, Vol. 9, No. 1, 2020, pp. 1-14.
- Barcellini, Wilma, et al. "New insights in autoimmune hemolytic anemia: From pathogenesis to therapy." Journal of Clinical Medicine, Vol. 9, No. 12, 2020, p. 3859.
- Salama, Abdulgabar. "Treatment options for primary autoimmune hemolytic anemia: A short comprehensive review." Transfusion Medicine and Hemotherapy, Vol. 42, No. 5, 2015, pp. 294-301.
- Yurek, Salih, et al. "Precautions surrounding blood transfusion in autoimmune haemolytic anaemias are overestimated." Blood Transfusion, Vol. 13, No. 4, 2015, pp. 616-21.
- Maral, Senem, et al. "Transfusion in autoimmune hemolytic anemia: Comparison of two different strategies." Medical Science and Discovery, Vol. 7, No. 8, 2020, pp. 589-93.
- Michalak, Sylwia Sulimiera, et al. "Autoimmune hemolytic anemia: Current knowledge and perspectives." Immunity & Ageing, Vol. 17, No. 1, 2020, pp. 1-16.