Research - International Journal of Medical Research & Health Sciences ( 2021) Volume 10, Issue 5
Effects of Pyroglutamyl Peptides on Depression, Anxiety and Analgesia in Mice
Oguz Mutlu1*, Pelin Tanyeri2, Seymanur Basarir2, Ibrahim Uygun1, Furuzan Akar1, Gulsen Celebi1, Mehmet Emin Buyukokuroglu2, Faruk Erden1 and Karel Vales32Pharmacology Department, Sakarya University, Sakarya, Turkey
3National Institute of Mental Health, Topolova, Klecany, Czech Republic
Oguz Mutlu, Pharmacology Department, Kocaeli University, Kocaeli, Turkey, Tel: +90 262 303 7250, Email: oguzmutlu80@hotmail.com
Received: 21-Apr-2021 Accepted Date: May 18, 2021 ; Published: 25-May-2021
Abstract
Objective: Insect Adipokinetic Hormones (AKH) play role in sugar and lipid mobilization and support the production of energy for the flight and movement of insects. In our previous studies, we showed that AKH had antidepressant, anxiolytic, and analgesic effects in mice and increased neurotrophic factors and neurogenesis in mice, and had beneficial effects in olfactory bulbectomy and posttraumatic stress disorder rat models. In our previous study, we hypothesized that metabolites of AKH, including pyroglutamic acid and pyroglutamyl peptides, pass the blood-brain barrier after intraperitoneal administration of AKH in rats, which is supported by other studies. Methods: In this study, we aimed to investigate the effects of three pyroglutamyl peptides on depression, anxiety, and analgesia in the Forced Swimming Test (FST), Elevated Plus Maze (EPM) test, and hot plate test in mice. We used L-pyroglutamic acid (pGlu; 50 mg/kg), pyroglutamic acid-valine (pGlu-Val; 10 mg/kg), pyroglutamic acid-leucine (pGlu-Leu; 10 mg/ kg) and pyroglutamic acid-valine-aspartate-phenylalanine (pGlu-Val-Asp-Phe; 10 mg/kg) as pyroglutamyl peptides. Results: In FST, all the peptides decreased immobility time, showing that these peptides had antidepressant-like effects. In the EPM test, none of the peptides had a significant effect, but tetrapeptide partially increased both % time spent in the open arm and % entry into the open arm. In the hot plate test, both dipeptides significantly increased latency of licking the arm showing that these dipeptides had analgesic effects. In the open field test, none of the studied peptides altered the total number of crossings compared to the control treatment. Conclusion: In conclusion, we found that pyroglutamyl peptides had preclinical beneficial effects on animal behavior in mice.
Keywords
Pyroglutamyl peptides, FST, EPM, Hot plate, Behaviour, Mice
Introduction
Depression and anxiety are widely seen disorders with significant morbidity and mortality. These disorders, which are caused by various stress factors and induce changes in neurotransmitter levels in the brain, also affect the structure and function of neuronal dendrites and cause histopathological changes in the volume and structure of important brain areas, such as the hippocampus, amygdala, prefrontal cortex, and striatum. These changes in the brain affect human behaviour and can lead to suicide attempts and disrupt learning and memory functions, including attention and motivation. The drugs available for the treatment of depression and anxiety can cause drug dependence, tolerance problems, and many side effects. Therefore, new strategies for the treatment of these disorders should be investigated.
Adipokinetic Hormones (AKHs) are metabolic neuropeptides that mediate the mobilization of energy substrates from the fat body in many insects [1]. These peptide hormones are products of neurosecretory neurons located in the corpora cardiaca, neuroendocrine glands attached to the insect brain. In our recent studies, we showed that the AKH/RPCH peptide family possesses antidepressant, anxiolytic and analgesic effects, causes hyperlocomotion, and exerts neuroprotective effects after chronic injection in mice [2]. Additionally, after acute injection, we observed antidepressant, anxiolytic, and analgesic effects of the AKH/RPCH family of peptides in mice [3]. In another study, we showed the promotive effects of these peptides on learning and memory in the MK-801-induced schizophrenia model in rats [4]. Finally, we found that these peptides relieved depression and anxiety and improved cognition in an olfactory bulbectomy model of depression and a posttraumatic stress disorder model in rats [5].
There are some structural and functional differences among the various types of insect AKHs. In our previous studies, we used three insect AKHs, the first of which was Anax imperator AKH (Ani-AKH), which had a sequence of pGlu- Val-Asn-Phe-Ser-Pro-Ser-Trp-NH2. The sequence of Libellula auripennis AKH (Lia-AKH) was determined to be pGlu-Val-Asn-Phe-Thr-Pro-Ser-Trp-NH2, and Phormia-Terra hypertrehalosemic hormone (Pht-HrTH) showed a sequence of pGlu-Leu-Thr-Phe-Ser-Pro-Asp-Trp-NH2 [6]. In our previous study, we hypothesized that metabolites of AKH, including pyroglutamic acid and pyroglutamyl peptides, may pass through the blood-brain barrier after intraperitoneal administration of AKH in rats, which is supported by other studies [7,8]. We thought that these metabolites are responsible for the behavioral effects of AKH seen in our previous studies [2-5]. For this reason, we studied the effects of L-pyroglutamic acid and pyroglutamyl peptides on depression, anxiety, and analgesia in this study. L-pyroglutamic acid was shown to both be converted into glutamate and antagonize the effects of glutamate in the brain [9]. Additionally, in recent studies, pyroglutamic acid reversed scopolamine-induced memory disturbance and increased cholinergic activity both in rodents and in humans [10,11]. Pyroglutamic acid also showed positive effects on memory in aged humans and animals [12]. In recent studies, food-derived pyroglutamyl peptides showed antidepressant and anxiolytic effects in mice [7,8].
This study aimed to investigate the effects of L-pyroglutamic acid and three pyroglutamyl peptides on depression, anxiety, and analgesia in the Forced Swimming Test (FST), Elevated Plus Maze test (EPM), and hot plate test in male balb-c mice. The locomotion of the animals was also investigated using the open field test. We used two dipeptides, pyroglutamic acid-valine (pGlu-Val) and pyroglutamic acid-leucine (pGlu-Leu), which were the starting sequences of the AKHs that we used in our previous studies [2-5]. We also investigated a tetrapeptide that has a sequence of pyroglutamic acid-valine-aspartate-phenylalanine (pGlu-Val-Asp-Phe) as a pyroglutamyl peptide.
Materials and Methods
Animals
Male inbred BALB/c ByJ mice (Sakarya University, Sakarya, Turkey) that were 7-8 weeks old upon arrival at the laboratory were kept in the laboratory for two weeks before the initiation of the experiments. The animals were maintained under standard laboratory conditions (12-h light: 12-h dark cycle, lights on at 07:00 h, T=21 ± 1°C). All animals received food and water ad libitum. All procedures described in this study were conducted by the European Community Council’s Directive for the Ethical Treatment of Animals (86/609/EEC) and with the approval of the Sakarya University Medical Faculty (27/2020).
Experimental Groups and Drug Administration
The effects of L-pGlu and pyroglutamyl peptides on depression, anxiety, analgesia, and locomotion were investigated in the FST, EPM test, hot plate test, and open field test, which are common and well-known tests used to evaluate depression, anxiety, analgesia, and locomotion, respectively. BALB/c mice were treated intraperitoneally and acutely with L-pyroglutamic acid (pGlu; 50 mg/kg; n=10), pyroglutamic acid-valine (pGlu-Val; 10 mg/kg; n=10), pyroglutamic acid-leucine (pGlu-Leu; 10 mg/kg; n=10), pyroglutamic acid-valine-aspartate-phenylalanine (pGlu-Val- Asp-Phe; 10 mg/kg; n=10) or vehicle (saline with 1% DMSO; n=10). All the drugs were given 30 min before the FST, EPM, hot plate, and open field tests in a volume of 0.1 ml/10 g body weight. The effective dose of each drug was selected according to previous behavioral and neurochemical studies [7,8]. L-pGlu was purchased from Sigma (St. Louis, USA), while pyroglutamyl peptides were synthesized in the chemistry department of UCT Prague.
Forced Swimming Test (FST)
The FST method used in this study was similar to a previously described method [13]. Briefly, the mice were placed individually into a Plexiglas cylinder (height: 25 cm, diameter: 10 cm) containing 10 cm of water maintained at 23°C-25°C and were left there for 6 min. Because this is a situation from which they cannot escape, the animals rapidly become immobile. They float in an upright position and make only small movements to keep their heads above water. The duration of immobility was recorded during the last 4 min of the 6 min testing period.
Elevated Plus Maze (EPM) Test
Anxiety-related behavior was measured by the EPM test. The experiments were conducted according to a previously described method [14]. Each mouse was placed at the center of the maze facing one of the open arms and was allowed to explore the maze. The open-arm activity was evaluated as follows: 1) the time spent in the open arms relative to the total time spent in the plus maze (300 s), expressed as a percentage and; 2) the number of entries into the open arms relative to the total number of entries into both the open and closed arms, expressed as a percentage. These values were used as indices of anxiety in mice.
Hot Plate Test
The hot plate test was used to measure the latency of the pain reaction [15]. The animals were placed into a glass square on a hot plate maintained at 55 ± 0.1°C. The latency to lick the hind paws or jump was recorded as an index of the pain reaction. A cutoff time of 60 s was used.
Open Field Test
Treatment effects on animal locomotor activity were measured using the open field test. This experiment was performed as previously described [16]. The apparatus consisted of a circular open field (height: 30 cm, diameter: 40 cm) made of PVC with a black-and-white-striped cardboard pattern (30 cm × 20 cm) nailed to one of the walls. The floor was divided into six peripheral sections and one central section of the same dimensions. A light bulb above the central section provided constant illumination of approximately 100×. The center zone of the open field was a circle that had a 12 cm diameter. To measure locomotor activity, an animal was placed in the center of the open field, and its behaviors were recorded for 5 min using the Etovision-XT video tracking system software (Noldus). The locomotor activity was evaluated by determining the number of crossings in seven sections of the apparatus.
Statistical Evaluation
The results of the FST, EPM, hot plate and open field tests were evaluated by one-way ANOVA followed by Tukey’s post hoc test when significant differences were detected. The data are expressed as the mean values ± SEM. The differences were considered statistically significant when p was less than 0.05.
Results
The Effects of Pyroglutamyl Peptides on Depression-like Behavior in the FST
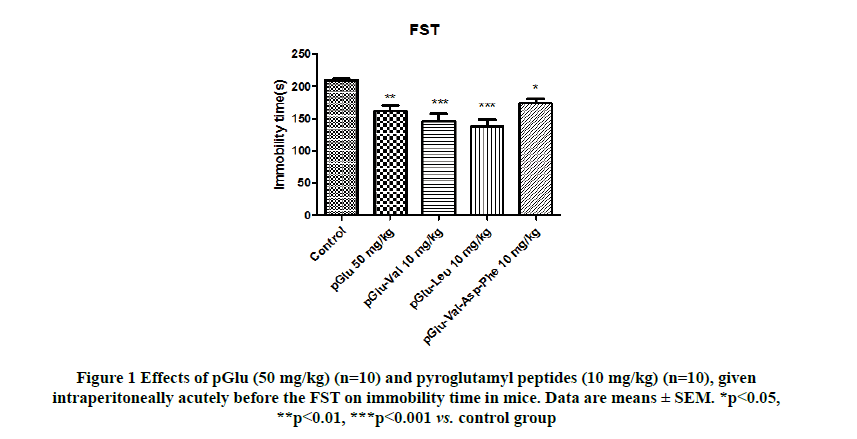
Significant differences were observed among the groups when the effects of pGlu (50 mg/kg) and pyroglutamyl peptides (10 mg/kg) on immobility time in the FST were evaluated (F(4,45)=10.81; p<0.0001; Figure 1). pGlu (p<0.01), pGlu- Val (p<0.001), pGlu-Leu (p<0.001), and pGlu-Val-Asp-Phe (p<0.05) significantly decreased the immobility time in the FST compared to the control treatment.
The Effects of Pyroglutamyl Peptides on Anxiety in the EPM test
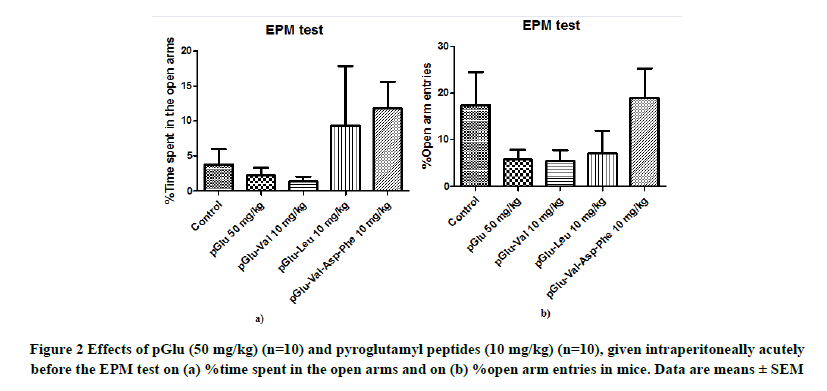
No significant differences were observed among the groups when the effects of pGlu (50 mg/kg) and pyroglutamyl peptides (10 mg/kg) on the % time spent in the open arms were evaluated in the EPM test (F(4,45)=1.125; p=0.35; Figure 2a). There were also no significant differences among the groups when the effects of pGlu (50 mg/kg) and pyroglutamyl peptides (10 mg/kg) on the % open arm entries were evaluated (F(4,45)=1.77; p=0.15; Figure 2b). pGlu- Val-Asp-Phe partially increased the % time spent in the open arms and the % open arm entries compared to the control treatment, but these differences were not significant.
The Effects of Pyroglutamyl Peptides on Analgesia in the Hot Plate Test
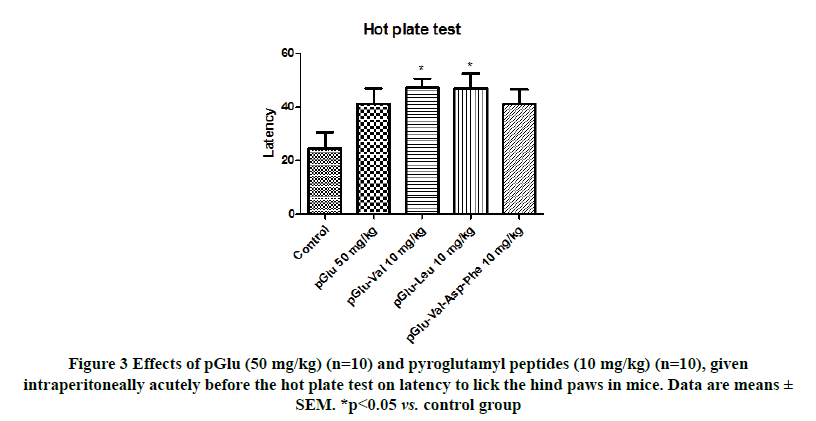
Significant differences were observed among the groups when the effects of pGlu (50 mg/kg) and pyroglutamyl peptides (10 mg/kg) on the latency to lick the hind paw or jump were evaluated in the hot plate test (F(4,45)=3.091; p<0.05; Figure 3). Both pGlu-Val (p<0.05) and pGlu-Leu (p<0.05) significantly increased the latency compared to the control.
The Effects of Pyroglutamyl Peptides on Locomotion in the Open Field Test
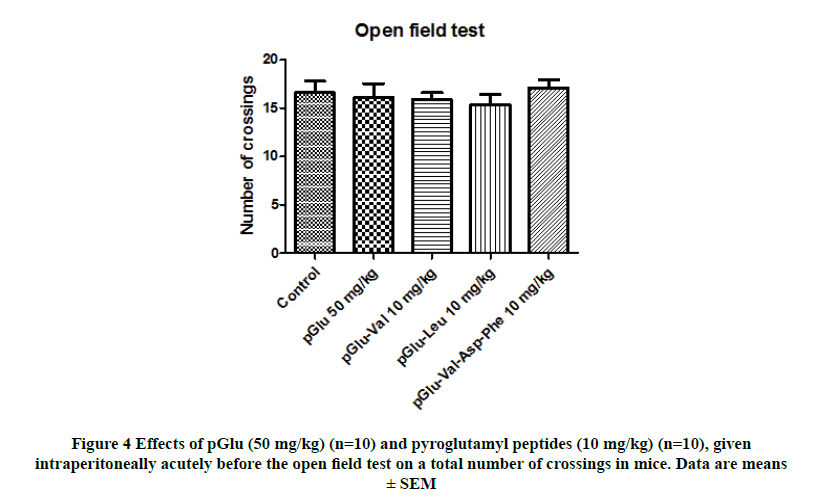
No significant differences were observed among the groups when the effects of pGlu (50 mg/kg) and pyroglutamyl peptides (10 mg/kg) on the total number of crossings were evaluated in the open field test (F(4,45)=0.38; Figure 4). None of the drugs altered the number of crossings compared to the control treatment.
Discussion
In this study, we found that pGlu (50 mg/kg) and pyroglutamyl peptides (10 mg/kg) decreased the immobility time in the FST, showing their antidepressant-like effects. pGlu-Val-Asp-Phe partially increased the % time spent in the open arms and the % open arm entries, while neither pGlu nor the other peptides showed any effect at the doses used. pGlu-Val and pGlu-Leu increased the latency in the hot plate test, showing their analgesic effects, while pGlu and tetrapeptide had no significant effects. None of the peptides changed locomotion at the doses used in the open field test.
Pyroglutamic acid (pGlu) is a cyclized derivative of L-glutamic acid. It is an uncommon amino acid derivative in which the free amino group of glutamic acid cyclizes to form a lactam ring. It is formed non-enzymatically from glutamate, glutamine, and gamma-glutamylated peptides, but it can also be produced by the action of gammaglutamylcyclotransferase on an L-amino acid. pGlu is sold over the counter as a “smart drug” for improving blood circulation in the brain. In previous studies, pGlu exerted beneficial effects on anxiety and memory in rodents [11,12,17]. In a recent study, oxiracetam and D-pyroglutamic acid antagonized the disruption of passive avoidance behavior induced by the N-methyl-D-aspartate receptor antagonist 2-amino-5-phosphonovalerate [18]. It was also shown that pGlu reversed scopolamine-induced memory impairment and affected brain cholinergic levels [10,11]. Pyroglutamic acid also relieved age-associated memory impairment in humans and improved learning and memory capacities in aged rats [12,19]. Food-derived pyroglutamyl peptides also showed anxiolytic and antidepressant-like effects in mice [7,8]. In our study, we used pGlu, which has been shown to have positive effects on behavior, as a reference drug and compared its effects with those of the pyroglutamyl peptides we used in this study.
Pyroglutamyl (pGlu) peptides are formed from the intramolecular cyclization of glutamine or glutamic acid residues at the N-terminal position of peptides. This process can occur endogenously or during the processing of foods containing the peptides. pGlu peptides are thought to have different characteristics, especially bitter and umami tastes, and thus can affect the sensory properties of foods that contain them. Moreover, some health-promoting properties have been reported for pGlu peptides, including hepatoprotective, antidepressant, and anti-inflammatory activities [20]. In our study, we used two dipeptides, pyroglutamic acid-valine (pGlu-Val) and pyroglutamic acid-leucine (pGlu- Leu), and a tetrapeptide with a sequence of pyroglutamic acid-valine-aspartate-phenylalanine (pGlu-Val-Asp-Phe) as pyroglutamyl peptides. These were the starting sequences of AKH that we used in our previous studies [2-5].
In our previous studies, we found antidepressant, anxiolytic, and analgesic effects of AKH in mice after both acute and chronic injection. In this study, we found that the pyroglutamyl peptides had antidepressant and analgesic effects, which was consistent with previous studies [7,8]. In this study, we did not detect a significant anxiolytic effect of pyroglutamyl peptides, which could have been been due to some nonspecific effects, such as laboratory conditions, although previous studies have also shown anxiolytic effects of pyroglutamyl peptides [8]. In our previous studies, we hypothesized that metabolites of AKH, including pyroglutamic acid and pyroglutamyl peptides, may pass through the blood-brain barrier after intraperitoneal administration of AKH in rats, which is supported by other studies [7,8]. In this study, we found that pyroglutamyl peptides had antidepressant and analgesic effects, supporting the hypothesis that pyroglutamyl metabolites of AKH may be responsible for the behavioral effects of AKH seen in our previous studies [2-5].
In our previous studies on AKH, we obtained variable results on locomotion. After chronic injection, we found that AKH enhanced locomotion, while acute injection of AKH decreased locomotion in mice [2,3]. In another study, we again observed increased motor activity with some AKH molecules in an olfactory bulbectomy rat model, while there was no effect on locomotion in a posttraumatic stress disorder rat model [5]. These differences in our previous studies may also be due to laboratory conditions, the different species of the animals used or different protocols, and different doses of drugs used. Effects on locomotion may alter the behavioral effects of investigated drugs. In this study, none of the peptides altered locomotion, so we can be sure that the results of this study are not due to nonspecific effects on locomotion.
In our previous study, we found that AKH is metabolized to smaller peptides in the blood circulation and that pyroglutamic acid (pGlu) and dipeptides, including pyroglutamic acid, may pass through the blood-brain barrier and may be responsible for the behavioral effects of AKH [5]. Additionally, in our previous study, we showed that the AKH/RPCH family of peptides reversed memory impairment in an MK-801-induced schizophrenia model, which suggests that these peptides may affect NMDA receptors [4]. L-pyroglutamic acid was shown to both be converted into glutamate and antagonize the effects of glutamate in the brain [9]. Additionally, in our previous study, we found that AKH increased brain dopamine levels in a posttraumatic stress disorder rat model [5]. Therefore, we hypothesize that pyroglutamyl peptides, which are active metabolites of AKH, may affect glutamatergic and dopaminergic systems, which may be responsible for the behavioral effects we observed in this study.
Conclusion
In conclusion, pyroglutamyl peptides seem to be responsible for the behavioral effects of AKH. These peptides also affect animal behavior after intraperitoneal administration, probably by affecting the dopaminergic and glutamatergic systems. Future in vitro and in vivo studies with different doses and different animal models should be performed to elucidate the exact mechanisms of the effects of these peptides.
Declarations
Conflicts of Interest
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
References
- Gade, G., and Heather G. Marco. "Adipokinetic and hypertrehalosemic neurohormones." Encyclopedia of Entomology, 2008, pp. 44-45.
- Mutlu, Oguz, et al. "Effects of chronic administration of adipokinetic and hypertrehalosemic hormone on animal behavior, BDNF, and CREB expression in the hippocampus and neurogenesis in mice." Fundamental and Clinical Pharmacology, Vol. 30, No. 1, 2016, pp. 4-13.
- Mutlu, Oguz, et al. "Effects of acute administration of adipokinetic hormone on depression, anxiety, pain, locomotion and memory in mice." Chinese Journal of Physiology, Vol. 60, No. 2, 2017, pp. 106-13.
- Mutlu, Oguz, et al. "Effects of the Adipokinetic Hormone/Red Pigment‐Concentrating Hormone (AKH/RPCH) family of peptides on MK‐801‐induced schizophrenia models." Fundamental and Clinical Pharmacology, Vol. 32, No. 6, 2018, pp. 589-602.
- Mutlu, Oguz, et al. "Effects of adipokinetic hormone/red pigment-concentrating hormone family of peptides in olfactory bulbectomy model and posttraumatic stress disorder model of rats." Peptides, Vol. 134, 2020, p. 170408.
- Gade, G. "The explosion of structural information on insect neuropeptides." Fortschritte der Chemie organischer Naturstoffe/Progress in the Chemistry of Organic Natural Products, Vol. 71, 1997, pp. 1-128.
- Yamamoto, Yukako, et al. "Antidepressant-like effect of food-derived pyroglutamyl peptides in mice." Neuropeptides, Vol. 51, 2015, pp. 25-29.
- Ohinata, K et al. "Antidepressant agent or anti-anxiety agent." Japan Patent 2014162735A.
- Kumar, Akhilesh, and Anand K. Bachhawat. "Pyroglutamic acid: Throwing light on a lightly studied metabolite." Current Science, Vol. 102, No. 2, 2012, pp. 288-97.
- Pepeu, Giancarlo, and Giacomo Spignoli. "Nootropic drugs and brain cholinergic mechanisms." Progress in Neuro-Psychopharmacology and Biological Psychiatry, Vol. 13, 1989, pp. S77-S88.
- Spignoli, G., et al. "Effect of pyroglutamic acid stereoisomers on ECS and scopolamine-induced memory disruption and brain acetylcholine levels in the rat." Pharmacological Research Communications, Vol. 19, No. 12, 1987, pp. 901-12.
- Drago, F., et al. "Pyroglutamic acid improves learning and memory capacities in old rats." Functional Neurology, Vol. 3, No. 2, 1988, pp. 137-43.
- Porsolt, R. D., A. Bertin, and M. J. A. I. P. Jalfre. "Behavioral despair in mice: a primary screening test for antidepressants." International Archives of Pharmacodynamics and Therapy, Vol. 229, No. 2, 1977, pp. 327-36.
- Lalonde, R., and C. Strazielle. "Relations between open-field, elevated plus-maze, and emergence tests as displayed by C57/BL6J and BALB/c mice." Journal of Neuroscience Methods, Vol. 171, No. 1, 2008, pp. 48-52.
- Duman, Erdem N., et al. "Effect of gender on antinociceptive effect of paroxetine in hot plate test in mice." Progress in Neuro-Psychopharmacology and Biological Psychiatry, Vol. 30, No. 2, 2006, pp. 292-96.
- Belzung, Catherine. "Measuring rodent exploratory behavior." Techniques in the Behavioral and Neural Sciences, Vol. 13, Elsevier, 1999, pp. 738-49.
- Beni, M., D. E. Pellegrini‐Giampietro, and F. Moroni. "A new endogenous anxiolytic agent: l‐pyroglutamic acid." Fundamental and Clinical Pharmacology, Vol. 2, No. 2, 1988, pp. 77-82.
- Paoli, F., G. Spignoli, and G. Pepeu. "Oxiracetam and D-pyroglutamic acid antagonize a disruption of passive avoidance behaviour induced by the N-methyl-D-aspartate receptor antagonist 2-amino-5-phosphonovalerate." Psychopharmacology, Vol. 100, No. 1, 1990, pp. 130-31.
- Grioli, S., et al. "Pyroglutamic acid improves the age associated memory impairment." Fundamental and Clinical Pharmacology, Vol. 4, No. 2, 1990, pp. 169-73.
- Gazme, Behzad, et al. "Occurrence, properties and biological significance of pyroglutamyl peptides derived from different food sources." Food Science and Human Wellness, Vol. 8, No. 3, 2019, pp. 268-74.