Research - International Journal of Medical Research & Health Sciences ( 2022) Volume 11, Issue 2
Comprehensive Review on Extracellular Matrix Metalloproteinase Inducer (EMMPRIN) with a Special Emphasis on Tumorigenesis in Oral Cancer
Sabiha Mokashi Khan1*, Nilima Prakash2 and Raiskhan Mokashi32Department of Oral Pathology and Microbiology, MGV KBH Dental College and Hospital, Nashik, Maharashtra, India
3Department of Conservative Dentistry and Endodontics, SMBT IDSR Dental College Dhamangaon, Nashik, Maharashtra, India
Sabiha Mokashi Khan, Oral Cancer Screening and Awareness in Cancer Screening Vertical, Indian Cancer Society, Mumbai, India, Email: dr.sabihamokashi@gmail.com
Received: 26-Jan-2022, Manuscript No. ijmrhs-22-52507 (M); Editor assigned: 31-Jan-2022, Pre QC No. ijmrhs-22-52507 (P); Reviewed: 05-Feb-2022, QC No. ijmrhs-22-52507 (Q); Revised: 18-Feb-2022, Manuscript No. ijmrhs-22-52507 (R); Published: 25-Feb-2022
Abstract
Extracellular matrix metalloproteinase inducer EMMPRIN also known as basigin or CD147 is a cell surface glycoprotein that belongs to the Ig superfamily. It is involved in numerous physiological and pathological processes and displays several molecular and cellular characteristics. The major function of EMMPRIN is to stimulate the synthesis of several matrix metalloproteinases that play an important role in the maintenance of the extracellular matrix. The role of the tumour microenvironment in oral carcinogenesis is taking centre stage. There is evidence that EMMPRIN is central in the promotion of tumour invasion, growth/progression, and metastasis of various cancers, and the mechanism by which it exerts its effect in Oral Squamous Cell Carcinoma (OSCC) tumorigenesis have not been completely understood. This article aims to comprehensively review the structure, biological function of EMMPRIN, and its role in oral tumorigenesis and possible therapeutic target.
Keywords
EMMPRIN, Structure, Biological function, MMP’s, OSCC tumorigenesis, Tumor growth and metastasis, Therapeutic target
Introduction
Extracellular matrix metalloproteinase inducer EMMPRIN also known as basigin or CD147 is a widely distributed cell surface glycoprotein that belongs to the Ig superfamily, it is involved in numerous physiological and pathological systems and display several molecular and cellular characteristics, but a major function of EMMPRIN is to stimulate the synthesis of several matrix metalloproteinases [1]. It was first labelled as ‘Tumour Cell-derived Collagenase Stimulatory Factor’ (TCSF) and later renamed ‘EMMPRIN’ to designate its role in extracellular matrix metalloproteinase induction via. normal, as well as pathological, cellular interactions [2]. EMMPRIN molecules are expressed at varying levels in multiple cell types, including epithelial cells, endothelial cells, and leukocytes [3]. It takes part in a variety of normal cell physiologies, including lymphocyte responsiveness and intracellular transportation [4]. EMMPRIN serves as a diagnostic and therapeutic target in cancer and inflammatory diseases, either directly or indirectly, by targeting EMMPRIN partners. It can trigger matrix metalloproteinase inductions involved in Extra Cellular Matrix (ECM) degradation, cell adhesion, and cell-cell interactions. It can also induce differentiation myofibroblast associated with ECM deposition and contraction. The shift from fibrosis to lysis, and vice versa, is not clearly understood and could involve EMMPRIN [5]. It is evidence that EMMPRIN is central in the promotion of tumour invasion, growth/progression, and metastasis as it upregulates MMPs secreted from adjacent fibroblasts [6]. Dysregulation of EMMPRIN has been associated with almost every type of cancer [7].
The significance of EMMPRIN in tumour growth and metastasis has been studied in detail, but the mechanisms by which it exerts its effects in OSCC tumorigenesis are not completely understood [8].
This comprehensive review will provide a detailed overview of EMMPRIN, its molecular structure, diverse biological functions of EMMPRIN, its role in OSCC tumorigenesis and therapeutic targets.
Structure and Functions of Emmprin
Structure of EMMPRIN
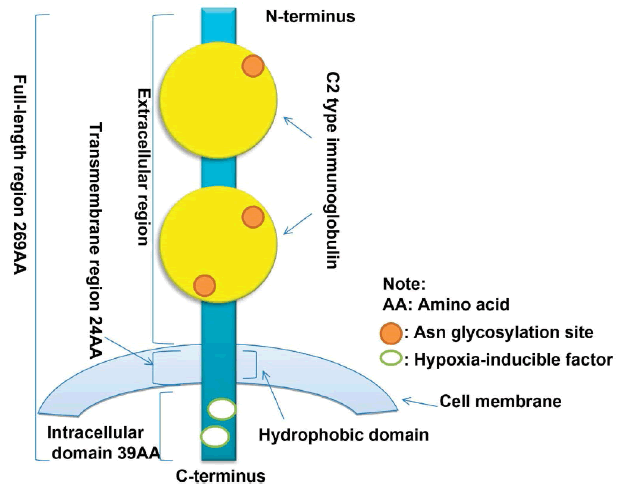
EMMPRIN or CD147 gene is localized to chromosome 19p13.3 and contains 1797 bp. The coding region of CD147 encodes 269 amino acid residues. The extracellular region of the N-terminal sequence has two C2-type immunoglobulin regions. 24 amino acid residues are in the transmembrane region and 39 amino acid residues are in the C-terminal intracellular region. There are two Hypoxia-Inducible Factor (HIF) binding sites in the 3’ flanking region of the CD147 gene. 21 highly conserved amino acid residues are present in the hydrophobic domain structure of the CD147 transmembrane region, which serve as both a signal peptide for CD147 and a cell membrane anchor. The presence of two immunoglobulin-like structures in the extracellular region of CD147 activates MMPs. Three Asn glycosylation sites are present in the CD147 extracellular region. Treatment of CD147 with glycosidase F will generate proteins with molecular masses ranging from 28,000 to 60,000 daltons, which indicates that the N-terminus of CD147 is highly glycosylated. Dimerization in the CD147 crystal structure plays an important role in letting CD147 take part in tumour cell invasion and MMP-2 production (Figure 1).
Figure 1. The molecular structure of EMMPRIN (CD147) [7]
Functions of EMMPRIN
EMMPRIN in organogenesis: EMMPRIN is expressed in both embryos and adults and plays fundamental roles in intercellular recognition involved in the differentiation and development of various cells, tissues, and organs as well as in various immunological phenomena. During organogenesis, EMMPRIN is expressed in epithelial tissues of various organs, brain ventricles, spinal cord, and dorsal root ganglion and concerning the skin; it is expressed in keratinocytes and is localized in the basal layer of the epidermis, suggesting the possibility that EMMPRIN may have a function in the embryonic development or differentiation of keratinocytes [9].
EMMPRIN neurological function: Extensive distribution of EMMPRIN in the brain plays an important role in learning and memory, as well as, in sensory functions. Lack of it may cause behavioural alterations [2].
EMMPRIN in reproduction: EMMPRIN is an important cell-surface molecule involved in early embryogenesis. Experimental evidence shows that knockout mice lacking the basigin gene (Bsg) mainly died around the time of implantation and as a small number of surviving adult mice were sterile, indicating that the EMMPRIN is important in development and reproduction [10].
EMMPRIN epithelial barrier function: EMMPRIN, by inducing MMP-9, is an important factor in the regulation of epithelial integrity. In corneal physiology and dry eye disease, EMMPRIN action on cell surface occludins is significant in maintaining corneal epithelium integrity [11].
EMMPRIN and interacting partners: EMMPRIN is demonstrated at varying levels in many cell types, including hematopoietic, epithelial, endothelial cells, and leukocytes. It has also been shown to regulate lymphocyte responsiveness, monocarboxylate transporter expression, and spermatogenesis. These functions reflect multiple interacting partners of EMMPRIN [12].
EMMPRIN as an inducer of MMPs: EMMPRIN stimulates the production of several matrix metalloproteinases such as MMP-1 (interstitial collagenase), MMP-2 (gelatinase A), MMP-3 (stromelysin 1), and MMP-9 (gelatinase B). It is not effective on their physiological inhibitors, TIMP-1 and TIMP-2, hence modifying the collagenolytic balance toward MMP production and activation. The role of EMMPRIN inducing MMP production is not limited to fibroblasts. EMMPRIN also had a paracrine effect on MMP production by endothelial cells, suggesting the potential implication of tumour-produced EMMPRIN in angiogenesis. Beyond its well-known paracrine effect, EMMPRIN induced MMPs within the same populations of cells, in both tumoral and nontumoral cells suggesting a more widespread role for EMMPRIN as an inducer of MMPs [13].
EMMPRIN expression in tumorigenesis: Cell invasion, metastasis, and angiogenesis are key factors for carcinogenesis. Dysregulation of EMMPRIN is reported in several types of cancer. It activates cell proliferation, invasion, and metastasis and inhibits tumour cell apoptosis under hypoxic conditions. EMMPRIN serves as a protein hub in cancer, as it is involved in several homophilic and heterophilic cellular interactions spanning the major hallmarks of cancer [14].
EMMPRIN regulates MMP and Vascular Endothelial Cell Growth Factor (VEGF) in tumour invasion and metastasis. It stimulates MMP production in stromal fibroblasts and endothelial cells around the tumour, as well as in tumour cells themselves, by a mechanism involving homophilic interactions between EMMPRIN molecules on opposing cells after membrane vesicle shedding. EMMPRIN stimulates angiogenesis via. induction of VEGF, invasiveness via. stimulation of MMP and multidrug resistance through hyaluronan mediated upregulation of ErbB2 signalling and cell survival pathway activities [12].
EMMPRIN plays a determinant role in mediating glycolysis for tumour growth, through functional cooperation with monocarboxylate transporter (MCT1 and MCT4) [15].
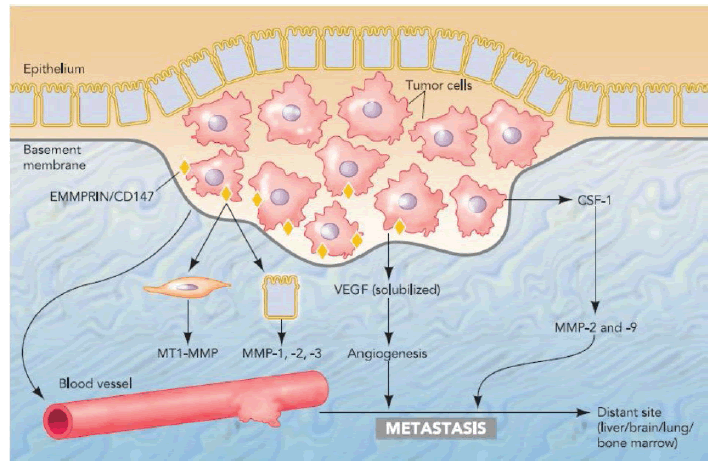
Malignant cells express EMMPRIN on their surface, which acts as a stimulator to produce MMP-1, -2, and -3 by endothelial cells and MT1-MMP by fibroblasts. Vascular Endothelial Growth Factor (VEGF) also induces the expression of several MMPs in endothelial cells. Tumour cells have recruited MMP-2 and -9, producing CD45- positive neutrophils and macrophages into the tumour tissue by the production of Colony-Stimulating Factor-1 (CSF- 1), which are then an abundant source of MMPs, which contribute to tumour progression (Figure 2) [16].
Figure 2. Interaction of Matrix metalloproteinase between epithelial and stromal cells in cancer and its role in tumour metastasis [16]
EMMPRIN contributes to cell adhesion modulation, tumour growth, invasion, and angiogenesis due to its association with several proteins implicated in different signalling pathways such as matrix metalloproteinases, ErbB, MAPK cascade proteins, monocarboxylate transporters (MCT), integrins, caveolin-1 (Cav-1), Tenascin (TN)-C, Vascular Endothelial Growth Factor (VEGF), urokinase-type plasminogen activator (uPA), and cyclophilins (Cyp) [17].
Increased expression of EMMPRIN in Oral squamous cell carcinoma has been reported to correlate with lymphatic metastasis and tumour progression. EMMPRIN upregulates urokinase-type plasminogen in oral tumours. The level of EMMPRIN determines the invasive potential through the regulation of the two proteolytic systems, MMPs and uPA; the increase in EMMPRIN expression during progression upregulates protease production and promotes greater invasion [8]. EMMPRIN protein is widely expressed in OSCC patients with a significant association with the degree of differentiation of the tumour [18]. Luís Silva Monteiro, et al., reveal that EMMPRIN protein is frequently overexpressed in OSCC, especially in high proliferative tumours, suggesting that it might be involved in the growth of these tumours [17]. Moderately and poorly differentiated tumours showed more EMMPRIN overexpression than well-differentiated tumours.
In OSCC, the prognostic value of EMMPRIN is poorly studied. More attention on EMMPRIN and its role in OSCC is needed, it can be used as an effective biological prognostic marker to identify high-risk OSCC patients.
EMMPRIN as a potential therapeutic target: Targeting tumour-associated antigens is fast emerging as an area of promise to treat late-stage and recurrent carcinomas. EMMPRIN is a multifunctional glycoprotein that can modify the tumour microenvironment by activating proteinases, inducing angiogenic factors in tumour and stromal cells, and regulating the survival and growth of anchorage-independent tumour cells (micrometastases) and Multidrug Resistance (MDR). It is involved in cancer drug resistance and plays a very important role in cancer metastasis. Thus, EMMPRIN may be ideal therapeutic targets to limit the progression and spread of cancer [19]. Blocking their activities is a possible strategy for controlling cancer spread. Although, clinical trials with synthetic MMP inhibitors have not yielded encouraging results to date. EMMPRIN could be a target because it causes overexpression of MMP in a cancer cell-dependent manner, through heterotypic cancer cell–fibroblast or homotypic cancer cell-cancer cell interactions. Moreover, the fact that EMMPRIN stimulates the growth and angiogenesis of tumours through the same cell interactions makes EMMPRIN more attractive as a target [2]. It has also been demonstrated that silencing EMMPRIN by RNA interference approach could inhibit tumour progression in murine lymphoid neoplasm and pancreatic cancer. It is also suggested that down-regulation of EMMPRIN by RNA interference could result in decreased X-linked inhibitors of apoptosis [20].
Conclusion
EMMPRIN is a highly glycosylated cell surface transmembrane protein involved in a wide range of processes, including angiogenesis, inflammatory diseases, and cancer progression. EMMPRIN could be used as an important biological prognostic marker to identify high-risk OSCC patients, which can help in making the right decision as to the appropriate treatment due to its multiple roles in cancer progression, including cell proliferation and vascularization. The precise understanding of how EMMPRIN activity is involved in tumorigenesis requires further study and its significant implications for cancer treatment.
Declarations
Conflict of Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
- Yan, Li, Stanley Zucker, and Bryan P. Toole. "Roles of the multifunctional glycoprotein, emmprin (basigin; CD147), in tumour progression." Thrombosis and Haemostasis, Vol. 93, No. 02, 2005, pp. 199-204.
Google Scholar Crossref - Nabeshima, Kazuki, et al. "Emmprin (basigin/CD147): Matrix metalloproteinase modulator and multifunctional cell recognition molecule that plays a critical role in cancer progression." Pathology International, Vol. 56, No. 7, 2006, pp. 359-67.
Google Scholar Crossref - Pata, Supansa, et al. "Differential CD147 functional epitopes on distinct leukocyte subsets." Frontiers in Immunology, Vol. 12, 2021, p. 3157.
Google Scholar Crossref - Huang, Zhiquan, et al. "Overexpression of EMMPRIN isoform 2 is associated with head and neck cancer metastasis." PLoS One, Vol. 9, No. 4, 2014, p. e91596.
Google Scholar Crossref - Guindolet, Damien, and Eric E. Gabison. "Role of CD147 (EMMPRIN/Basigin) in tissue remodeling." The Anatomical Record, Vol. 303, No. 6, 2020, pp. 1584-89.
Google Scholar Crossref - Yang, Min, et al. "Prognostic significance of CD147 in patients with glioblastoma." Journal of Neuro-oncology, Vol. 115, No. 1, 2013, pp. 19-26.
Google Scholar Crossref - Xiong, Lijuan, Carl K. Edwards, and Lijun Zhou. "The biological function and clinical utilization of CD147 in human diseases: A review of the current scientific literature." International Journal of Molecular Sciences, Vol. 15, No. 10, 2014, pp. 17411-41.
Google Scholar Crossref - Lescaille, Géraldine, et al. "EMMPRIN/CD147 up-regulates urokinase-type plasminogen activator: Implications in oral tumor progression." BMC Cancer, Vol. 12, No. 1, 2012, pp. 1-9.
Google Scholar Crossref - Chen, Xiang, Takuro Kanekura, and Tamotsu Kanzaki. "Expression of Basigin in human fetal, infantile and adult skin and in basal cell carcinoma." Journal of Cutaneous Pathology, Vol. 28, No. 4, 2001, pp. 184-90.
Google Scholar Crossref - Igakura, Tadahiko, et al. "Roles of basigin, a member of the immunoglobulin superfamily, in behavior as to an irritating odor, lymphocyte response, and blood-brain barrier." Biochemical and Biophysical Research Communications, Vol. 224, No. 1, 1996, pp. 33-36.
Google Scholar Crossref - Huet, Eric, et al. "EMMPRIN modulates epithelial barrier function through a MMP-mediated occludin cleavage: Implications in dry eye disease." The American Journal of Pathology, Vol. 179, No. 3, 2011, pp. 1278-86.
Google Scholar Crossref - Yurchenko, Vyacheslav, Stephanie Constant, and Michael Bukrinsky. "Dealing with the family: CD147 interactions with cyclophilins." Immunology, Vol. 117, No. 3, 2006, pp. 301-09.
Google Scholar Crossref - Huet, Eric, et al. "Role of emmprin/CD147 in tissue remodeling." Connective Tissue Research, Vol. 49, No. 3-4, 2008, pp. 175-79.
Google Scholar Crossref - Kumar, Dhivya, et al. "Structural insights on druggable hotspots in CD147: A bull's eye view." Life Sciences, Vol. 224, 2019, pp. 76-87.
Google Scholar Crossref - Le Floch, Renaud, et al. "CD147 subunit of lactate/H+ symporters MCT1 and hypoxia-inducible MCT4 is critical for energetics and growth of glycolytic tumors." Proceedings of the National Academy of Sciences, Vol. 108, No. 40, 2011, pp. 16663-68.
Google Scholar Crossref - Sengupta, N., and T. T. MacDonald. "The role of matrix metalloproteinases in stromal/epithelial interactions in the gut." Physiology, Vol. 22, No. 6, 2007, pp. 401-09.
Google Scholar Crossref - Monteiro, Luís Silva, et al. "EMMPRIN expression in oral squamous cell carcinomas: Correlation with tumor proliferation and patient survival." BioMed Research International, Vol. 2014, 2014.
Google Scholar Crossref - Sahaf, Rakia, et al. "Immunohistochemical expression of EMMPRIN (CD147) in oral squamous cell carcinoma." Journal of Oral and Maxillofacial Surgery, Medicine, and Pathology, Vol. 30, No. 1, 2018, pp. 67-73.
Google Scholar Crossref - Hao, J. L., et al. "CD147/EMMPRIN and CD44 are potential therapeutic targets for metastatic prostate cancer." Current Cancer Drug Targets, Vol. 10, No. 3, 2010, pp. 287-306.
Google Scholar Crossref - Tian, Li, et al. "EMMPRIN is an independent negative prognostic factor for patients with astrocytic glioma." PloS One, Vol. 8, No. 3, 2013, p. e58069.
Google Scholar Crossref